Expanding the repertoire of optogenetic controlB6;129B6(Cg)-Actb<tm1(tetO-ChR2*C128S/EYFP)Kftnk>/KftnkRbrc RBRC05454
Courtesy of Kenji F. Tanaka, M.D., Ph.D., Keio University |
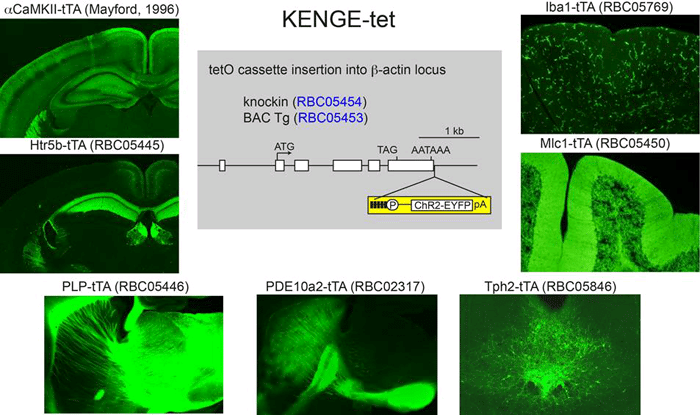
KENGE-tet enables us to achieve sufficient opsin expression to trigger photocurrents in combination with existing cell type-specific tTA lines. All pictures show tTA-mediated ChR2-EYFP induction by KENGE-tet.
Optogenetic strategies for controlling neuronal activity have been employed widely for mapping functional circuits within the nervous system. Channelrhodopsin-1 (ChR1) and ChR2, related microbial-type rhodopsins from Chlamydomonas reinhardtii, are involved in the generation of photocurrents. ChR2 is a light-gated cation channel, whereas ChR1 is a light-gated proton channel. The N-terminal 315 amino acids of ChR2 are homologous to the seven trans-membrane structure of numerous microbial-type rhodopsins. Inward currents in ChR2-expressing cells can be evoked after a flash of blue light in the presence of all-trans retinal [1, 2].
Drs. Tanaka and Yamanaka established the Knockin-mediated ENhanced Gene Expression by improved tetracycline-controlled gene induction (KENGE-tet) system for reliable generation of transgenic mice with light-responsive cells. They achieved this through targeting transgene insertion just downstream of the β-actin gene polyadenylation signal and through generating tetO-ChR2(C128S)-EYFP knock-in mice by homologous recombination of embryonic stem cells. They then demonstrated that ChR2(C128S)-mediated inward currents resulted in c-fos mRNA induction in astrocytes, showing that the KENGE-tet system is applicable to the optogenetic manipulation of both excitable neurons and non-excitable glial cells [3]. The results show that their system is applicable not only to neuroscience but also to any biomedical study requiring an understanding of how the activity of a selected population of cells is propagated through intricate organic systems.
| Related strain | : | B6(Cg)-Tg(tetO-ChR2*C128S/EYFP)1Kftnk/KftnkRbrc RBRC05453 | |
| Depositor | : | Kenji F. Tanaka, M.D., Ph.D. Department of Neuropsychiatry, Keio University School of Medicine Akihiro Yamanaka, Ph.D. Research Institute of Environmental Medicine, Nagoya University |
|
| References | : | [1] | Nagel G, Szellas T, Huhn W, Kateriya S, Adeishvili N, Berthold P, Ollig D, Hegemann P, Bamberg E. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci U S A.; 100(24):13940-5, 2003. |
| [2] | Boyden ES, Zhang F, Bamberg E, Nagel G, Deisseroth K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci.; 8(9):1263-8, 2005. | ||
| [3] | Tanaka KF, Matsui K, Sasaki T, Sano H, Sugio S, Fan K, Hen R, Nakai J, Yanagawa Y, Hasuwa H, Okabe M, Deisseroth K, Ikenaka K, Yamanaka A. Expanding the repertoire of optogenetically targeted cells with an enhanced gene expression system. Cell Rep.; 2(2):397-406, 2012. | ||