2. High-Speed Congenics Using First-Wave
Male Germ Cells
The fastest generation turnover in mammals (40-45 days)
|

Immature sperm (round spermatids) in the dotted circle (right) from a 22-day old male (black mouse) can be used in high-speed breeding of genetically mouse models
|
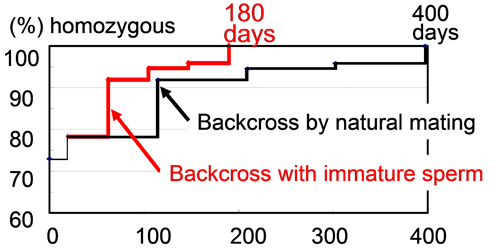
Time course of generation turnover and the rate of markers for the expected genetic background. With immature sperm, all markers became homozygous at 180 days in this case.
|
| In laboratory mice and rats, congenic breeding is essential for analyzing the genes of interest on specific genetic backgrounds and for analyzing quantitative trait loci. However, in theory it takes about 3-4 years to achieve a strain carrying about 99% of the recipient genome at the tenth backcrossing (N10). The recently developed‘speed congenic strategy’ using marker-assisted selection has shortened the required generations to N4 or N5 but it still takes more than a year. To accelerate the congenic breeding, we examined the feasibility of the microinsemination (sperm injection) technique using the “first wave” sperm cells from immature males. Haploid germ cells (round spermatids) first appear in immature male mice at 18 days after birth and their number increases to the practical level for microinsemination at 22-25 days [1]. The technique was then applied to the congenic strategy in combination with marker-assisted selection. Transgenic, knock-in, and ENU-mutant strains with mixed genetic backgrounds eventually became C57BL/6 congenic strains according to the polymorphic markers used (n= 74 to 176) [2]. The strategy was proven to be effective for other genetic backgrounds (e.g., NOD-scid). It should provide the fastest protocol for precise definition of the phenotypic effects of genes of interest on desired genetic backgrounds. |
|
Collaborated with Dr. H. Kurihara (Graduate School of Medicine, University of Tokyo, Japan)
|




