Fluorescent labeling of GABAergic neurons in the GAD67-GFP knock-in mouseICR.Cg-Gad1<tm1.1Tama>/Rbrc (RBRC03674)
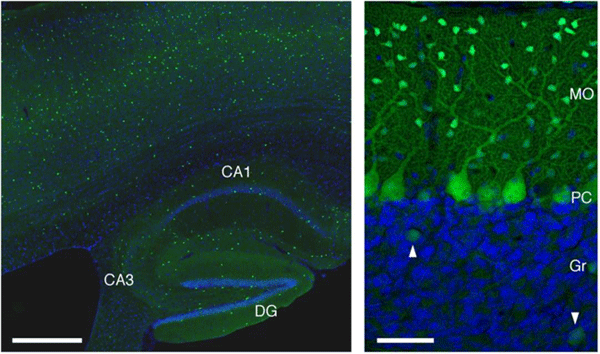
Courtesy of Yuchio Yanagawa, M.D. Expression of EGFP in the GAD67-GFP knock-in mouse. (Left) Fluorescent photomicrograph of a sagittal section of the GAD67-GFP knock-in mouse brain (4 weeks old) shows scattered distribution of EGFP in the hippocampus and cerebral cortex. (Right) Fluorescent photomicrograph of a sagittal section of the GAD67-GFP knock-in mouse cerebellar cortex (4 weeks old). Arrowheads in the right panels show Golgi cells. Merged images include nuclear staining with DAPI (blue). CA1-3, CA1-3 fields of Ammon’s horn; DG, dentate gyrus; Gr, granule cell layer of cerebellar cortex; MO, molecular layer of cerebellar cortex; PC, Purkinje cell layer. Scale bars: left, 500 µm; right, 50 µm. Photographs were provided by Dr. Ryosuke Kaneko, Gunma University. |
| Depositor | : | Yuchio Yanagawa, M.D. Department of Genetic and Behavioral Neuroscience Gunma University Graduate School of Medicine |
|
| References | : | [1] | Tamamaki N, Yanagawa Y, Tomioka R, Miyazaki J, Obata K, Kaneko T. Green fluorescent protein expression and colocalization with calretinin, parvalbumin, and somatostatin in the GAD67-GFP knock-in mouse. J Comp Neurol.; 467(1):60-79, 2003. |
| [2] | Kaneko K, Tamamaki N, Owada H, Kakizaki T, Kume N, Totsuka M, Yamamoto T, Yawo H, Yagi T, Obata K, Yanagawa Y. Noradrenergic excitation of a subpopulation of GABAergic cells in the basolateral amygdala via both activation of nonselective cationic conductance and suppression of resting K+ conductance: A study using glutamate decarboxylase 67-green fluorescent protein knock-in mice. Neuroscience 157, 781-797, 2008. | ||
| [3] | Brown RE, McKenna JT, Winston S, Basheer R, Yanagawa Y, Thakkar MM, McCarley RW. Characterization of GABAergic neurons in rapid-eye-movement sleep controlling regions of the brainstem reticular formation in GAD67-green fluorescent protein knock-in mice. Eur J Neurosci.; 27(2):352-63, 2008. | ||
| [4] | Hirono M, Saitow F, Kudo M, Suzuki H, Yanagawa Y, Yamada M, Nagao S, Konishi S, Obata K. Cerebellar globular cells receive monoaminergic excitation and monosynaptic inhibition from Purkinje cells. PLoS One 7:e29663, 2012. | ||
| [5] | Kawabata I, Kashiwagi Y, Obashi K, Ohkura M, Nakai J, Wynshaw-Boris A, Yanagawa Y, Okabe S. LIS1-dependent retrograde translocation of excitatory synapses in developing interneuron dendrites. Nat Commun. 3:722, 2012. | ||