A tool for the conditional deletion of the transcriptional repressor Bcl6B6.Cg-Bcl6/Rbrc RBRC05663
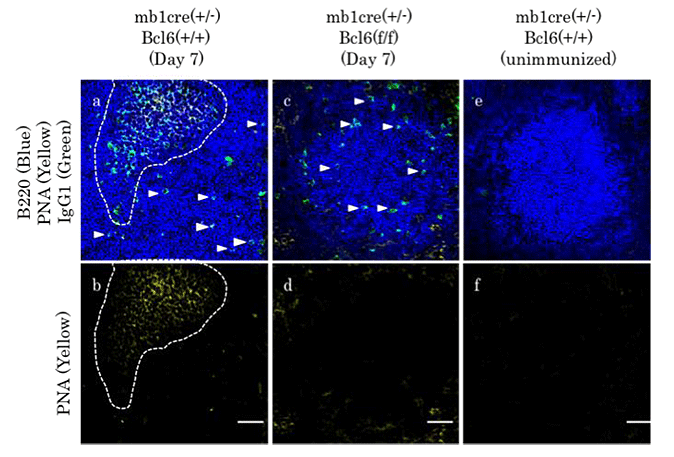
Courtesy of Toshitada Takemori, M.D., Ph.D., RIKEN Center for Integrative Medical Sciences Histological distribution of IgG1+ memory B cells in the spleens of conditional Bcl6-deficient (Bcl6f/f x mb-1-cre+/-; c and d) and control (Bcl6+/+ x mb-1-cre+/-; a and b) mice by confocal microscopic analysis. IgG1+ cells (green) within the B220+ follicles (blue) were identified from the images, and their expression of peanut agglutinin (PNA, yellow) was determined. IgG1+ cells of the B220+PNA-phenotype (memory cells) were located within the B cell follicles at day 7 after immunization in mutant (c and d) and control (a and b) mice. IgG1+ cells of the plasma cell phenotype were confined largely to the extrafollicular areas (data not shown). |
| Depositor | : | Toshitada Takemori, M.D., Ph.D. RIKEN Center for Integrative Medical Sciences |
| Reference | : [1] | Kaji T, Ishige A, Hikida M, Taka J, Hijikata A, Kubo M, Nagashima T, Takahashi Y, Kurosaki T, Okada M, Ohara O, Rajewsky K, Takemori T. Distinct cellular pathways select germline-encoded and somatically mutated antibodies into immunological memory. J. Exp. Med.; 209(11):2079-97, 2012. |