|
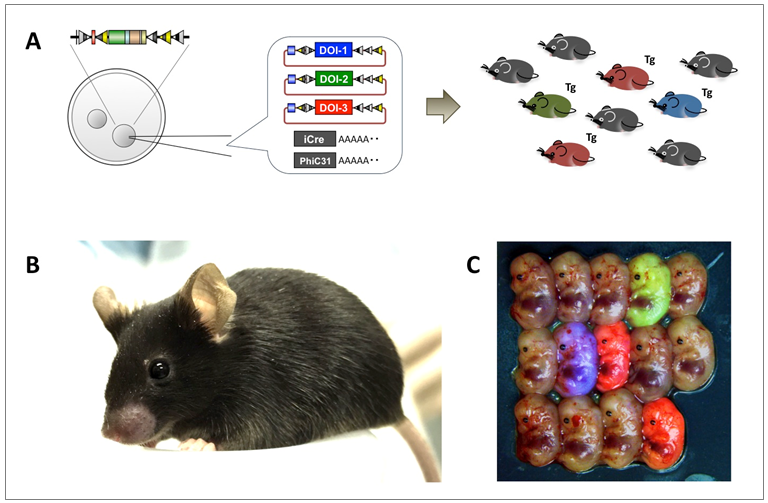
One-step generation of multiple transgenic mouse lines C57BL/6-Gt(ROSA)26Sor<tm10(PITT)Maoh> (RBRC06517)Courtesy of Masato Ohtsuka, Ph.D. Generation of multiple targeted transgenic mouse lines using i-PITT. (A) Schematic of simultaneous production of multiple transgenic lines using i-PITT. Multiple donor vectors that harbor different DNA of interest (DOI) are mixed and co-injected with iCre and PhiC31o mRNA into the fertilized eggs carrying the i-PITT landing pad in their genome. Appearance of different fluorescent colors indicates successful insertion of DOI. (B) TOKMO-3 mouse. (C) Day 13.5 fetuses derived from injected zygotes showing an example of simultaneous production of multiple targeted transgenics. Fetuses showing blue, green or red fluorescence indicate successful insertion of DOI from mCFP, eGFP or tdTomato vectors respectively. |
| Transgenic mice have been a valuable bioresource for studying gene function in vivo, although traditional strategy has certain drawbacks as a result of randomness of copy number and insertion site of the transgene. In order to target a single-copy transgene to a specific locus, Ohtsuka and colleagues generated a “seed mice strain” containing a landing pad at Rosa26 locus to apply novel technique named pronuclear injection-based targeted transgenesis (PITT) [1-5]. They have recently developed an improved-PITT (i-PITT) method by generating new seed mice strain (called TOKMO-3) with a C57BL/6N genetic background, in which Rosa26 locus was targeted with a combination of elements including loxP derivatives, FRT derivatives, and attP sites [6]. Insertion efficiencies in the mouse ES cells and zygotes were increased by combining Cre-loxP and PhiC31-attP/B system. They produced up to three transgenic strains in a single microinjection session by co-injecting multiple donor vectors with iCre and PhiC31o mRNAs into zygotes. The i-PITT system offers a new tool for transgenic technology which has a comparable efficiency to the emerging knockout technology such as CRISPR/Cas9 system. |
| Depositor | : | Masato Ohtsuka, Ph.D. Department of Molecular Life Science Tokai University School of Medicine |
|
| Strain name | : | C57BL/6-Gt(ROSA)26Sor<tm10(PITT)Maoh> | |
| RBRC No. | : | RBRC06517 | |
| References | : | [1] | Ohtsuka M, Ogiwara S, Miura H, Mizutani A, Warita T, Sato M, Imai K, Hozumi K, Sato T, Tanaka M, Kimura M, Inoko H. Pronuclear injection-based mouse targeted transgenesis for reproducible and highly efficient transgene expression. Nucleic Acids Res.; 38(22):e198, 2010. |
| [2] | Ohtsuka M, Miura H, Nakaoka H, Kimura M, Sato M, Inoko H. Targeted transgenesis through pronuclear injection of improved vectors into in vitro fertilized eggs. Transgenic Res.; 21(1):225-6, 2012. | ||
| [3] | Ohtsuka M, Miura H, Sato M, Kimura M, Inoko H, Gurumurthy CB. PITT: Pronuclear Injection-based Targeted Transgensis, a Reliable Transgene Expression Method in Mice. Exp. Anim.; 61(5):489-502, 2012. | ||
| [4] | Ohtsuka M, Miura H, Hayashi H, Nakaoka H, Kimura M, Sato M, Gurumurthy CB, Inoko H. Improvement of pronuclear injection-based targeted transgenesis (PITT) by iCre mRNA-mediated site-specific recombination. Transgenic Res.; 22(4):873-5, 2013. | ||
| [5] | Ohtsuka M. Development of pronuclear injection-based targeted transgenesis in mice through Cre-loxP site-specific recombination. Methods Mol Biol.; 1194:3-19, 2014. | ||
| [6] | Ohtsuka M, Miura H, Mochida K, Hirose M, Hasegawa A, Ogura A, Mizutani R, Kimura M, Isotani A, Ikawa M, Sato M, Gurumurthy CB. One-step generation of multiple transgenic mouse lines using an improved Pronuclear Injection-based Targeted Transgenesis (i-PITT). BMC Genomics; 16:274, 2015. | ||
| September 2015 Contact: Shinya Ayabe, Ph.D. Experimental Animal Division, RIKEN BioResource Center All materials contained on this site may not be reproduced, distributed, displayed, published or broadcast without the prior permission of the owner of that content. |