|
August 2017 Mouse of the Month |
Transcranial imaging of intracellular Ca2+ in astrocytes and neurons
C57BL/6-Tg(Slc1a2-G-CaMP7)Bsi (RBRC09650) |
|
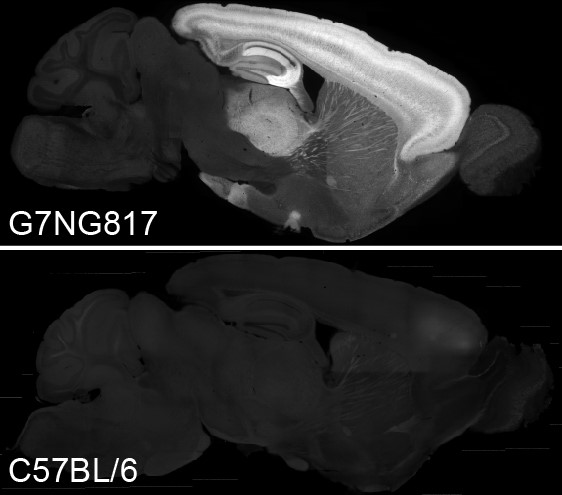
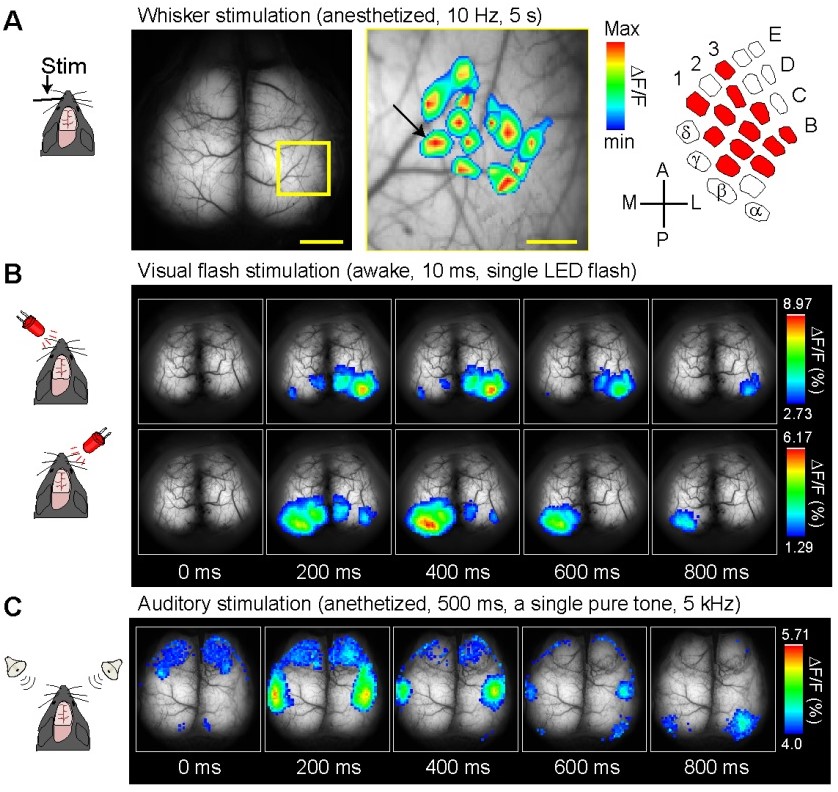
Transcranial direct current stimulation (tDCS) is a non-invasive treatment, in which weak direct current is applied through the skull. tDCS has been known to ameliorate neuropsychiatric and neurological conditions in humans. Although membrane potential fluctuations of astrocytes, the major glia cell type in the brain, are within a few millivolts from resting potential, recent reports have raised the possibility that intracellular Ca2+ elevation in astrocytes results in gliotransmission. Hirase and colleagues sought to reveal cellular mechanism for tDCS-induced plasticity in the mouse brain. They generated the G7NG817 mouse line that expresses G-CaMP7, the improved variant of genetically encoded calcium indicator G-CaMP [1], under a GLT-1 (or Slc1a2). The G7NG817 mouse was found to express high levels of G-CaMP7 in astrocytes and a subpopulation of neurons in the forebrain including the cortex and hippocampus, allowing transcranial Ca2+ imaging with a standard fluorescence microscope. Using G7NG817 mice, they showed that tDCS results in an enhancement of sensory evoked cortical response via astrocytic Ca2+ surges mediated by alpha-1 adrenergic receptor signaling [2, 3]. Recently, G7NG817 mice were used to characterize the in vivo functional dynamics of the somatosensory cortex in autistic mice [4], demonstrating the versatility of this mouse line by cross-breeding with various disease model mice. |
| Depositor | : | Hajime Hirase, Ph.D. Laboratory for Neuron-Glia Circuitry RIKEN Brain Science Institute |
|
| Strain name | : | C57BL/6-Tg(Slc1a2-G-CaMP7)Bsi | |
| RBRC No. | : | RBRC09650 | |
| References | : | [1] | Ohkura M, Sasaki T, Sadakari J, Gengyo-Ando K, Kagawa-Nagamura Y, Kobayashi C, Ikegaya Y, Nakai J. Genetically encoded green fluorescent Ca2+ indicators with improved detectability for neuronal Ca2+ signals. PLOS ONE; 7(12):e51286, 2012. |
| [2] | Monai H, Ohkura M, Tanaka M, Oe Y, Konno A, Hirai H, Mikoshiba K, Itohara S, Nakai J, Iwai Y, Hirase H. Calcium imaging reveals glial involvement in transcranial direct current stimulation-induced plasticity in mouse brain. Nat Commun.; 7:11100, 2016. | ||
| [3] | Monai H, Hirase H. Astrocytic calcium activation in a mouse model of tDCS-Extended discussion. Neurogenesis (Austin); 3(1):e1240055, 2016. | ||
| [4] | Nakai N, Nagano M, Saitow F, Watanabe Y, Kawamura Y, Kawamoto A, Tamada K, Mizuma H, Onoe H, Watanabe Y, Monai H, Hirase H, Nakatani J, Inagaki H, Kawada T, Miyazaki T, Watanabe M, Sato Y, Okabe S, Kitamura K, Kano M, Hashimoto K, Suzuki H, Takumi T. Serotonin rebalances cortical tuning and behavior linked to autism symptoms in 15q11-13 CNV mice. Sci Adv; 3(6):e1603001, 2017 | ||
| “Calcium imaging reveals glial involvement in transcranial direct current stimulation-induced plasticity in mouse brain.” Monai H, Ohkura M, Tanaka M, Oe Y, Konno A, Hirai H, Mikoshiba K, Itohara S, Nakai J, Iwai Y, Hirase H. | |
| Nat Commun.; 7:11100, 2016. https://doi.org/10.1038/ncomms11100 |
 |
| August 2017 Contact: Shinya Ayabe, Ph.D. Experimental Animal Division, RIKEN BioResource Center All materials contained on this site may not be reproduced, distributed, displayed, published or broadcast without the prior permission of the owner of that content. |