A tool for the generation of germline-competent iPS cellsSTOCK Tg(Nanog-GFP,Puro)1Yam RBRC02290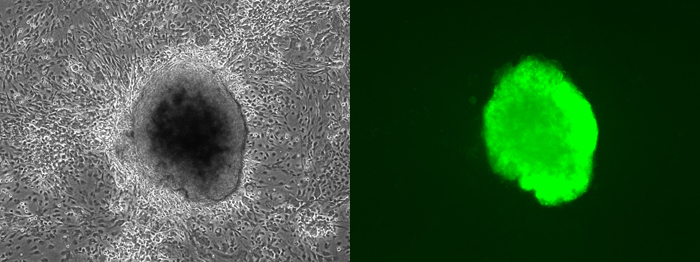 Germline-competent iPS cells from a Nanog-GFP transgenic mouse. The Nanog gene is a good candidate marker for the maintenance of pluripotent differentiation because of the loss of pluripotent epiblasts in Nanog knockout mice, the tendency of Nanog-null embryonic stem (ES) cells to differentiate spontaneously, and the capability of Nanog-overexpressing ES cells to retain self-renewal independently of leukemia inhibitory factor. The Nanog-GFP transgene was generated by the insertion of a GFP-IRES-puromycin resistance gene (Puror) cassette into the 5′ untranslated region of a bacterial artificial chromosome containing the mouse Nanog gene. ES cells with the Nanog-GFP transgene were positive for GFP, but became negative when differentiation was induced. With the use of this system, pluripotent stem cells competent for germline chimeras can be induced from Nanog-GFP transgenic mouse fibroblasts by the introduction of candidate genes for pluripotency. |
The 2012 Nobel Prize in Physiology or Medicine was awarded jointly to Sir John B. Gurdon and Dr. Shinya Yamanaka “for the discovery that mature cells can be reprogrammed to become pluripotent” [1]. In 2006, Dr. Yamanaka and colleagues generated induced pluripotent stem (iPS) cells from embryonic and adult fibroblasts in mice by the transfection of four factors: Oct3/4, Sox2, Klf4, and c-Myc [2]. In 2007, his group reported that a similar approach was applicable to human fibroblasts, and generated human iPS cells [3]. A modified method is also available to induce iPS cells from wild-type mouse fibroblasts in the absence of antibiotic selection, as well as by using only three factors, without c-Myc, or by continual transfection of plasmid vectors [4-6]. Human iPS cells can be generated using episomal plasmid vectors encoding L-MYC, LIN28, and p53 shRNA, replacing c-MYC and NANOG [7].
Dr. Yamanaka has kindly deposited his mice and iPS cells at RIKEN BioResource Center. The list of deposited mouse strains is shown here. For iPS cells derived from laboratory mice and humans, please contact RIKEN BioResource Center Cell Bank.
| Depositor | : | Dr. Shinya Yamanaka Director of Center for iPS Cell Research and Application (CiRA) Kyoto University |
| References | : | [1] | The Nobel Prize in Physiology or Medicine 2012 – Novel Media AB http://www.nobelprize.org/nobel_prizes/medicine/laureates/2012/ |
| [2] | Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell; 126(4):663-76, 2006. | ||
| [3] | Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell; 131(5):861-72, 2007. | ||
| [4] | Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature; 448(7151):313-7, 2007. | ||
| [5] | Nakagawa M, Koyanagi M, Tanabe K, Takahashi K, Ichisaka T, Aoi T, Okita K, Mochiduki Y, Takizawa N, Yamanaka S. Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nat Biotechnol; 26(1):101-6, 2008. | ||
| [6] | Okita K, Hong H, Takahashi K, Yamanaka S. Generation of mouse-induced pluripotent stem cells with plasmid vectors. Nat Protoc; 5(3):418-28, 2010. | ||
| [7] | Okita K, Matsumura Y, Sato Y, Okada A, Morizane A, Okamoto S, Hong H, Nakagawa M, Tanabe K, Tezuka K, Shibata T, Kunisada T, Takahashi M, Takahashi J, Saji H, Yamanaka S. A more efficient method to generate integration-free human iPS cells. Nat Methods; 8(5):409-12, 2011. |





