A Dnmt1 knock-out mouse for epigenetic studies129;B6-Dnmt1tm1Okan (Acc. No. CDB0500K) RBRC04774
|
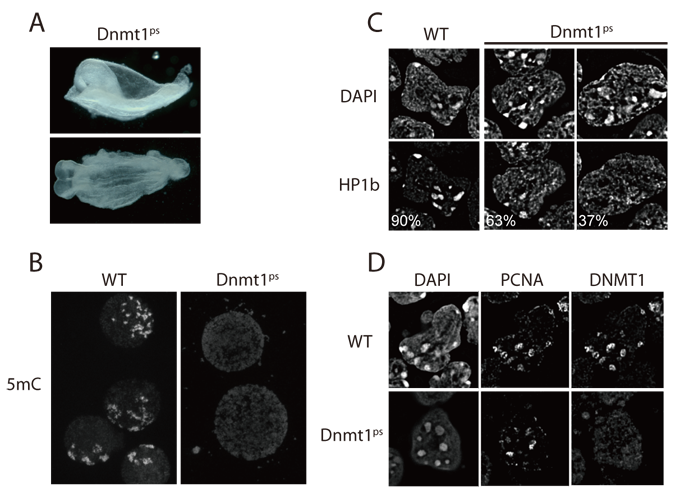
Epigenetic changes are defined as modifications that induce heritable changes in gene expression without changes in the DNA sequence. The methylation of cytosine on the 5-carbon position is a heritable epigenetic DNA modification that affects mammalian development. Three DNA methyltransferases, DNMT1, DNMT3A, and DNMT3B, are coordinately expressed and play an important role in the regulation of gene expression, genomic imprinting, and embryogenesis. 129;B6-Dnmt1tm1Okan mice (Acc. No. CDB0500K, RBRC04774) were generated to examine the roles of the catalytic activity of DNMT1 in vivo by creating a Dnmt1psallele that expresses a protein with a point mutation (DNMT1-C1229S) that lacks catalytic activity. Homozygous Dnmt1psmutant mice exhibit developmental arrest, accompanied by multiple defects, shortly after gastrulation. The phenotypic similarity of the homozygous Dnmt1psmutant mice with DNMT1-null mutant (Dnmt1c) mice clearly indicates that the catalytic activity of DNMT1 is essential for mammalian development. The 129;B6-Dnmt1tm1Okan mice together with other Dnmt knock-out mice are invaluable strains to study the role of these methyltransferases in mammalian epigenetics.
| Other related strains | : | B6;129S4-Dnmt1tm2Enl RBRC01737 B6;129S4-Dnmt3atm1Enl RBRC03730 B6;129S4-Dnmt3atm3.1Enl RBRC03731 B6;129S4-Dnmt3btm1Enl RBRC03732 B6;129S4-Dnmt3btm5.1Enl RBRC03733 |
|
| Depositor | : | Dr. Masaki Okano, RIKEN Center for Development Biology | |
| Reference | : | Takebayashi S, Tamura T, Matsuoka C, Okano M. Major and essential role for the DNA methylation mark in mouse embryogenesis and stable association of DNMT1 with newly replicated regions. Mol Cell Biol. 2007 27(23):8243-58. | |